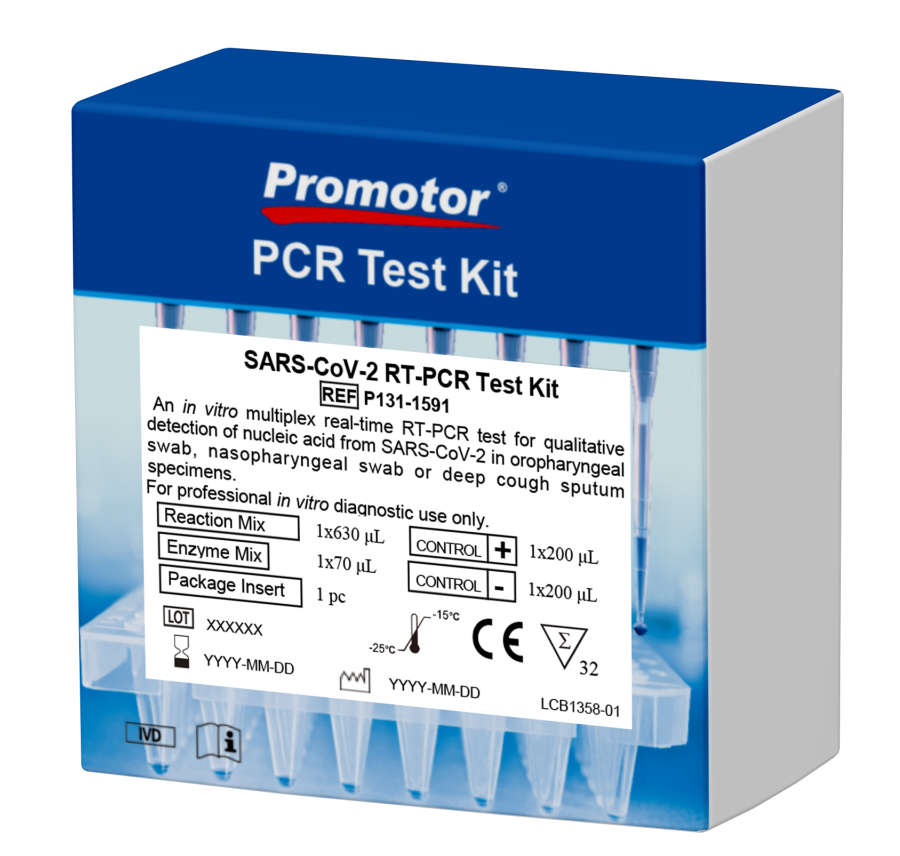
SARS-CoV-2 RT-PCR Test Kit

Product is Available for International Distribution Only – Not Available in the US
Promotor® PCR Test Kits
SARS-CoV-2 RT-PCR Test Kit
Qualitative detection of nucleic acid from SARS-CoV-2 in oropharyngeal swab, nasopharyngeal swab and deep
cough sputum specimens collected from individuals suspected of SARS-CoV-2 by their healthcare provider. *
*Clinical correlation with patient history and other diagnostic information is necessary to determine patient infection status.

Advantages and performance
Specimens include
Oropharyngeal and nasopharyngeal swabs, and deep cough sputum specimens
Qualitative detection of nucleic acids from SARS-CoV-2
Target Regions
N, E, ORF1ab
High Accuracy
97.93 % (95 % CI: 95.56% – 99.24 %)
Full-process controls with negative, positive and internal controls
Store at - 20 ± 5 ºC
CE Marked
Promotor® SARS-CoV-2 RT-PCR Test Kit
Product Specifications
| Feature | Specification |
| Technology | Real-Time reverse transcription polymerase chain reaction (rRT-PCR) |
| Target Region | N, E, ORF1ab |
| Specimen Type | Oropharyngeal swab, nasopharyngeal swab and deep cough sputum |
| Internal Control | Yes |
| Minimum Sample Volume | 200 μL |
| Test Duration | 90 minutes |
| Limit of Detection | 500 copies/mL |
| Precision of In-batch and batch (CV) | < 5 % |
| Positive Percent Agreement | 97.46 % (95 % CI: 92.75 % – 99.47 %) |
| Negative Percent Agreement | 98.26 % (95 % CI: 94.99 % – 99.64 %) |
| Accuracy | 97.93 % (95 % CI: 95.56% – 99.24 %) |
| Storage Temperature | – 20 ± 5 ºC |
| Package Size | 32 Tests/Kit |
Resource Library
Browse our Resources Library for documents available for download.
